Generally, transition metal catalysts are essential for the reductive hydrogen atom transfer reaction, also known as the transfer hydrogenation reaction or the borrowing-hydrogen reaction. It has been reported that graphene can be an active catalyst in ethylene and nitrobenzene reductions, but no report has described carbon-based materials as catalysts for alcohol amination via the borrowing-hydrogen reaction mechanism. The group headed by Prof. SHI Feng at the Lanzhou Institute of Chemical Physics (LICP) of the Chinese Academy of Sciences (CAS) has realized the transition metal-free reductive hydrogen atom transfer transformation using a carbon material as the catalyst with alcohol amination as well as nitro compound and ketone reduction as model reactions. 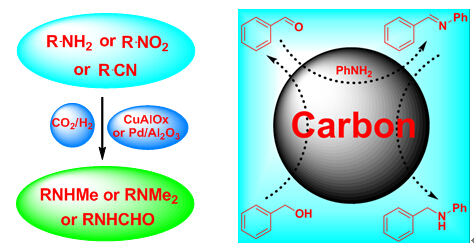
Carbon-catalysed reductive hydrogen atom transfer reactions (Image by SHI Feng et al.) The carbon catalyst exhibited excellent catalytic performance and good generality in the above reactions. The catalyst/product separation was easily performed, and the carbon catalyst was recyclable for several runs without observable deactivation. The results suggested that the C=O group may be the catalytically active site in this catalyst, while a high surface area of the carbon catalyst is important for achieving high activity. The activity of the base-activated carbon catalyst remained unchanged during the reuse process due to its stable structure. This new catalytic system may provide an attractive and useful methodology for a wider range of applications for industrially important compounds and intermediates. The work has been published in Nature Communications(2015, 6, 6478, doi: 10.1038/ncomms7478) . |
