The hydroformylation of alkenes is recognized as one of the most momentous and atom-economical forms of access to aldehydes, which are widely applied as intermediates for the synthesis of esters, alcohols, carboxylic acids, aliphatic amines and other fine chemicals. Nevertheless, one main challenge in the heterogeneous hydroformylation of alkenes is how regioselectivity should be controlled.
Recently, a research group led by Professor CUI Xinjiang and Professor SHI Feng from the Lanzhou Institute of Chemical Physics (LICP) of the Chinese Academy of Sciences (CAS) developed a novel catalyst for the regioselective heterogeneous hydroformylation of alkenes.
Based on the concept of bridging homogeneous and heterogeneous catalysis, the researchers synthesized a one-pot encapsulation of single-atom Rh catalysts within porous monophosphine polymers for the regioselective heterogeneous hydroformylation of alkenes, yielding 92% linear aldehyde selectivity (Fig. 1). In addition, this catalyst exhibited excellent catalytic activity (at least 60,000) and good stability.
This study was published in Journal of Catalysis on August 9, 2021.
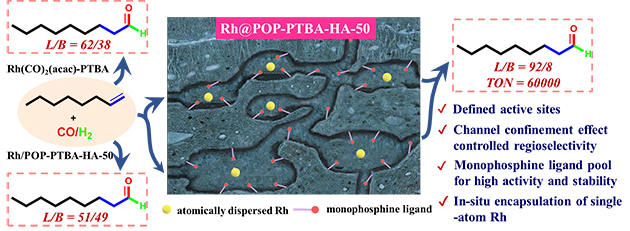
Fig. 1. Conceptual design for designing heterogeneous catalysts in regioselective hydroformylation.
Detailed characterization indicated that single-atom Rh catalysts were uniformly encapsulated within a porous monophosphine polymer. The researchers found that the encapsulation of Rh species within confined POPs exhibited better regioselectivity for the alkenes hydroformylation reaction than the polymer-loaded Rh catalyst. They revealed that encapsulation of atomically dispersed Rh catalysts within porous monophosphine polymers may endow the catalysts with high regioselectivity, whereas the encirclement of metal-active species by abundant but dispersive phosphine ligands strengthens the stability of the catalysts through multiple-site coordination.
These findings provide meaningful insights for designing heterogeneous catalysts with good regioselectivity, as well as for merging the advantages of homogeneous and heterogeneous catalysis.
This work was supported financially by the National Natural Science Foundation of China, the National Key Research and Development Program of China and the Youth Innovation Promotion Association of CAS.
Contact:
Wang Hongli
Email: wanghl@licp.cas.cn
Lanzhou Institute of Chemical Physics


