Abstract
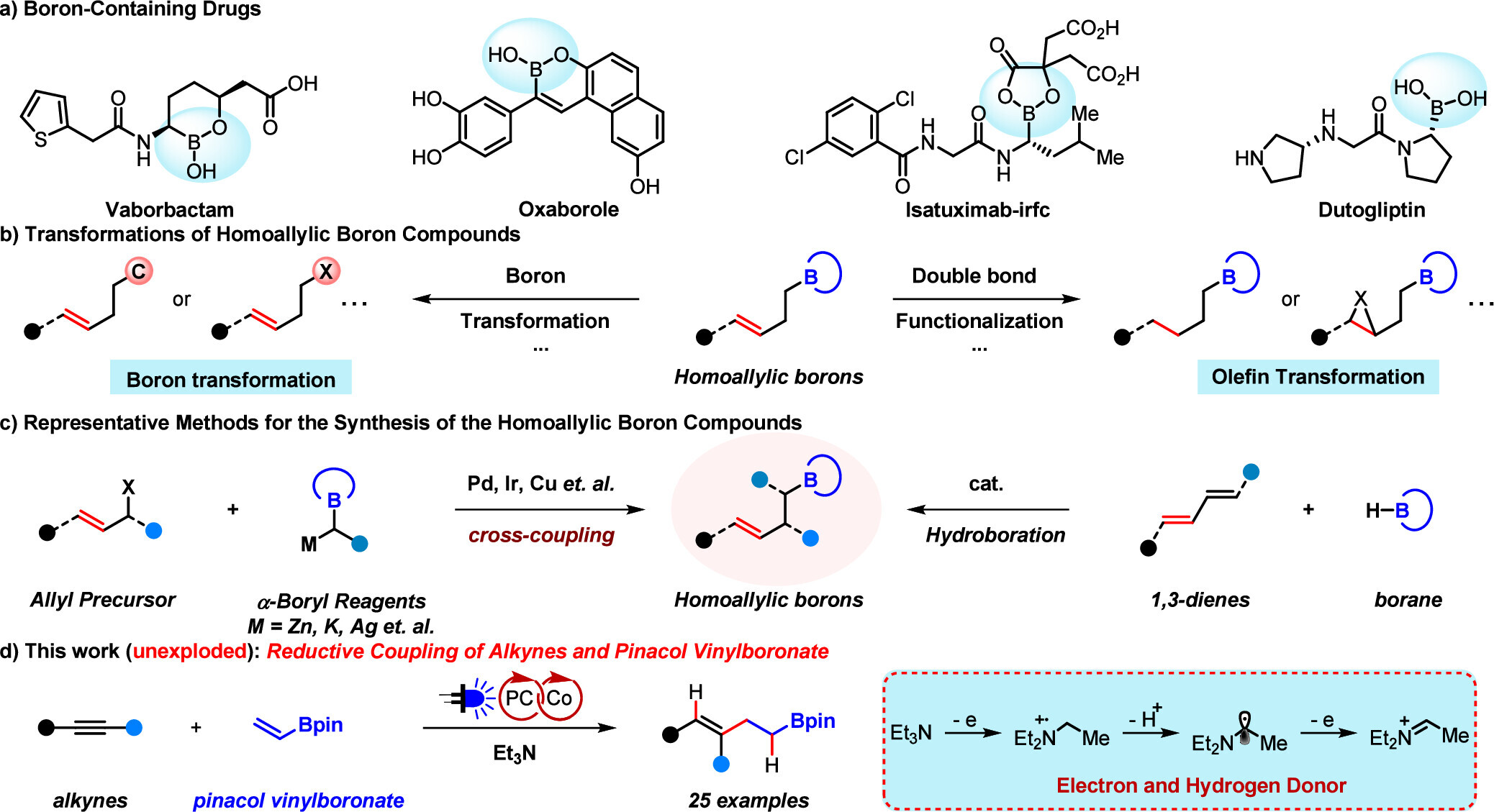
While conventional methods for constructing homoallylic boron scaffolds face limitations in pre-functionalization requirements and selectivity control, we herein disclose a visible-light-driven photoredox cobalt-catalyzed system that enables direct synthesis of multisubstituted homoallylic boron units through reductive coupling of vinyl boron reagents and alkynes. Utilizing triethylamine as both an electron and hydrogen donor, this strategy provides a step-economical approach with mild conditions and broad functional group tolerance, facilitating access to valuable organoboron building blocks.
Keywords Plus: ALLYLATION,ARYLATION,ENYNES,BORON,ACIDS
Published in ORGANIC LETTERS,Volume28;10.1021/acs.orglett.5c04880,FEB 27 2026


