Abstract
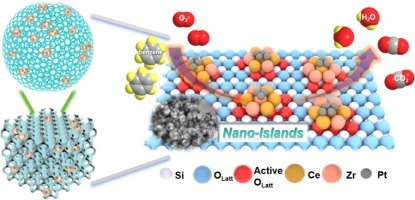
In this work, a novel and facile nano-islands Pt/CeZr/3DOM SiO2 catalyst was successfully synthesized. It was discovered that this catalyst had a strong oxygen activation ability and tunable metal-support interaction, achieving efficient low-temperature benzene oxidation. Systematic characterizations showed that when the calcination temperature was 800 degrees C, the nano-islands formed a strong interaction with the 3DOM SiO2 support. Meanwhile, introducing Zr species greatly changed the size and properties of the nano-islands, resulting in smaller and more numerous morphologies. This provided abundant sites for anchoring Pt species. The presence of oxygen vacancies and confinement of Zr species promoted the dispersion and anchoring of Pt species. Oxygen vacancies also accelerated the activation of surface oxygen molecules, promoting benzene oxidation. Mechanism studies revealed that in the Pt/Ce7Zr7-Ce10Zr10/Si-800 catalyst, stronger Pt-Ce interaction and more active Pt-cluster species could adsorb and activate benzene, converting it into intermediates that react with active oxygen, leading to improved low-temperature benzene oxidation performance. This work provides new insights for designing efficient Pt nano-island catalysts for low-temperature benzene oxidation.
Keywords Plus: CO OXIDATION,PERFORMANCE,REDUCTION,ZRO2,NO
Published in APPLIED SURFACE SCIENCE,Volume725;10.1016/j.apsusc.2026.165834,APR 15 2026


