Abstract
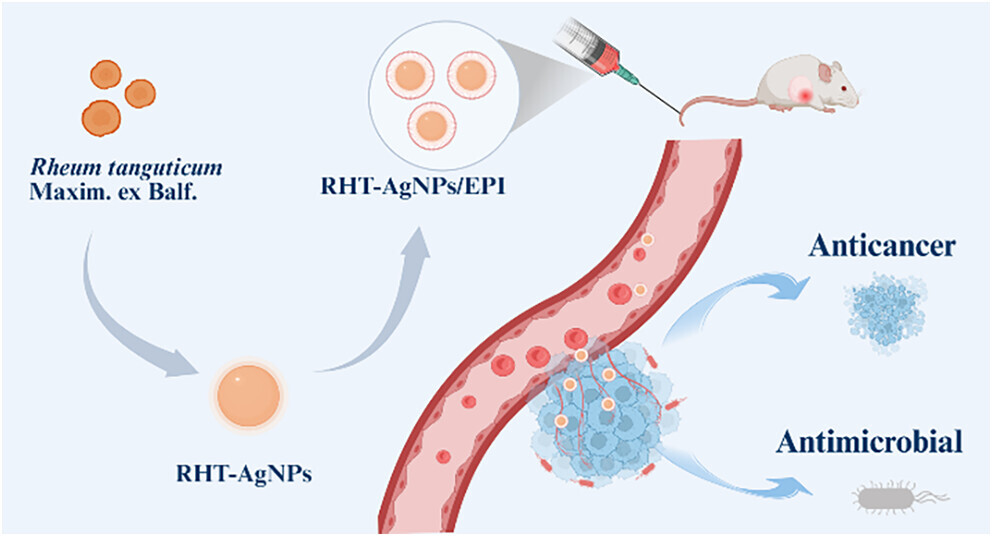
Antibacterial nanocarrier-based anticancer drug delivery systems have garnered significant attention in the treatment of bacteria-associated cancers. By employing natural polyphenols in a green synthesis process, the inert surface of conventional silver nanoparticles (AgNPs) can be modified to enable anticancer drug loading and provide dual antibacterial and anticancer functionalities. However, the development of AgNPs with intrinsic antibacterial and antitumor activities for anticancer drug loading and bacteria-associated tumor combination therapy has not been extensively explored. Here, the extract of Rheum tanguticum Maxim. ex Balf. (RHT), an important traditional Chinese medicine, was utilized as a reducing and stabilizing agent for the green synthesis of AgNPs (RHT-AgNPs). The resultant RHT-AgNPs had spherical morphology, good dispersion, uniform particle size (15.14 +/- 0.73 nm), and remarkable long-term stability in aqueous solutions (>43 days). Mass spectra (MS) and high-performance liquid chromatography (HPLC) analysis were performed to identify the main constituents in rhubarb extract responsible for the preparation of RHT-AgNPs. The resultant RHT-AgNPs (500 mu g/mL) exhibited low long-term (72 h) cytotoxicity against normal cells (cell viability >54%) and retained significant antibacterial activity against both Escherichia coli and Staphylococcus aureus. More importantly, the RHT-AgNPs exhibited significant cytotoxic activity against breast cancer cells (IC50 = 77.92 mu g/mL), which originated from the rhubarb extract (IC50 = 20.36 mu g/mL), thus enabling an enhanced antitumor effect with the loaded anticancer agent. RHT-AgNPs demonstrated high drug loading efficiency (>86%) for anticancer drug epirubicin (EPI), and the resultant EPI-loaded RHT-AgNPs (RHT-AgNPs/EPI) nanoformulation exhibited unique pH- and glutathione (GSH)-responsive EPI release as well as pH-responsive Ag release behavior. The in vitro cytotoxicity assay indicated that RHT-AgNPs/EPI could significantly improve the effect of bacteria on the cytotoxicity of EPI against breast cancer cells (with an equivalent EPI concentration of 20 mu g/mL). Moreover, in an S. aureus infection-associated 4T1 breast tumor-bearing Balb/c mouse model, intravenous administration of RHT-AgNPs/EPI (with an equivalent EPI amount of 5 mg/kg) effectively suppressed infectious inflammation and showed superior tumor suppression compared to the single EPI administration without inducing notable toxic effects on healthy tissues.
Keywords Plus: TUMOR,ANTHRAQUINONES,NANOPARTICLES,MICROBIOME,EPIRUBICIN,MECHANISMS,NANOSILVER,DRUGS,SIZE
Published in ACS APPLIED MATERIALS & INTERFACES,Volume18;10.1021/acsami.5c23737,FEB 18 2026


